Lung cancer is known for its aggressiveness. In recent years, new hopes have arisen for patients with the advent of immunotherapy, a new treatment approach consisting in stimulating the patient’s own immune system. In lung cancer, this passes through the administration of specific drugs targeting immune checkpoint inhibitors (ICI). ICIs stop the interactions between cancer cells and specific proteins present in immune cells that prevent the latter from recognizing the cancer cells as harmful – the idea is to switch the immune system “back on”, help it identify cancerous cells and in turn fight them more effectively.
The idea came about after 2018, when James Allison, head of the department of immunology at the Anderson Cancer Center in Houston, and Tasuku Honjo, head of the department of immunology and genomic medicine at Kyoto University, were awarded the Nobel Prize for discoveries that paved the way for immunotherapy to fight advanced cancer.
Lung cancer (and in particular non-small cell lung cancer – NSCLC) immunotherapy has had success mainly through the two ICIs that are PD-1 and PDL-1 inhibitors, but it is important to note that (1) their efficacy could be better (2) not all patients respond equally to the treatment, for reasons still not well clarified. These are the core unmet needs that the I3LUNG project, and INT as a participating center, want to respond to provide an answer to. Specifically, by creating a dataset made up of different biomarkers (a meta-biomarker) that when put together can accurately predict a patient’s response to immunotherapy. These markers will be identified by looking at the results of the clinical studies conducted within the broader I3LUNG research project, whose data we will analyze with the help of Artificial Intelligence models designed ad hoc for this task.
In the first year of the project, aside from coordinating the project the Istituto Nazionale dei Tumori di Milano (INT) has been active in steering the design and creation of an original and unique electronic database to input and analyze clinical, epidemiological, molecular and radiological data at multiple levels, that has been populated with the data from over 1500 (so far) NSCLC patients collected by the clinical partners of the project. In parallel to the collection of retrospective data, the INT has designed and kick-started a prospective study enrolling 200 patients from five cancer centers, whose data will be collected and analyzed according to the AI methodologies we will implement from the autumn of 2023 onwards. The institute will be responsible for a set of clinical and biological analyses as well, in particular the analysis of patients’ microbiota and microRNA.
This project is already having a positive impact at a larger level at the Institute, with the constitution of a multidisciplinary team comprising physicians, bioengineers, nurses, biologists, data managers, pharmacists and project managers. This has been especially useful to create links, integrate the different skills and coordinate the progress of the project in the best possible way. A strong interaction is also being built with other related projects, such as the Apollo 11 network, 40+ Italian cancer centers that will share data through harmonized AI procedures, and share samples through an innovative “virtual biobank” design.
Spurred by this project, the idea of INT setting up Italy’s first Artificial Intelligence in Oncology Laboratory has emerged. It will be concretized with its creation in 2023 in the Institute’s Cascina Rosa Campus, already home to the its Data Science department.
Article written by the Istituto Nazionale dei Tumori di Milano (INT). For more information, please visit the Project section in our website or contact us.
On June 30th and July 1st, this year’s I3LUNG Annual Meeting took place at the MEDSIR offices in Barcelona, Spain. The venue offered stunning 360º views of the city. As we did last year, the meeting brought together the 16 partners of the project to share the progress achieved over the past year.
The event kicked off on Friday morning with a workshop in collaboration with Lung Cancer Europe focused on ‘Knowledge on Artificial Intelligence applied to treatment decision-making‘. This workshop was attended by 6 people in person and one attendee remotely, including lung cancer patients and patient advocates.
AnteriorAfter the workshop, we enjoyed lunch on the 27th floor in the Open Space of MEDSIR offices. It provided a great opportunity for the workshop participants and the I3LUNG consortium to mingle, chat and have a wonderful time all together. In addition, a photographer took pictures of those who posed in front of the specially designed photocall for the event.
Following the lunch, the Annual Meeting began with a warm welcome from Susana Vitorino, MEDSIR Trial & Client Manager, followed by Arsela Prelaj, PI of the I3LUNG, along with Laura Mazzeo, from the Istituto Nazionale dei Tumori di Milano (INT). The agenda for the first day was divided into six parts:
During the breaks on the first day, we conducted interviews with speakers from each institution, asking them two questions about the project. These interviews are intended to be published on the website, keep tuned!
We ended the first day presentations with a guest, Dr. Denis Horgan, who gave a talk about “Innovation and incentives: power for civilization to tackle lung cancer –and for better healthcare for ALL”. It was a pleasure to have him with us!
To conclude the day, we gathered at the Barcelona Milano restaurant and we enjoyed ourselves.
The second day began at 9 a.m. with an agenda divided into seven parts:
And we concluded the event with a small snack, providing an opportunity to bid farewell to the attendees.
Looking forward to next year’s Annual Meeting in Greece to analyse the progression made. Feel free to reach out to the people involved to know better about the I3LUNG!

The Integrative science, Intelligent data platform for Individualized LUNG cancer care with Immunotherapy (I3LUNG) consortium, funded by Horizon Europe, is leveraging artificial intelligence (AI) methods to develop a clinical decision-making tool for personalized lung cancer care. The goal is to improve the matching of metastatic non-small cell lung cancer (mNSCLC) patients with the most suitable immunotherapy (IO) or non-IO treatments. This can potentially revolutionize care by improving treatment outcomes, reducing unnecessary costs, and ensuring that patients most unlikely to benefit from IO therapy are provided more appropriate treatments alternatives and are spared the risk of serious IO-related adverse events like hyperprogression.
The Swedish Institute for Health Economics continues to develop the economic models that will be used support economic analysis of the I3LUNG AI platform from the many perspectives used in participating countries including stakeholders in the EU member states, the US, and Israel.
The conceptualization and programming of a budget impact model has been successfully completed. This model will play a vital role in informing stakeholders about the short-term budgetary implications and any budget reallocations that may be required in conjunction with adoption of the I3LUNG tool. The model incorporates intentional flexibility to consider the diverse perspectives of stakeholders in different settings. Our next step is to incorporate the results of the I3LUNG study when they become available and then to utilize the model empirically.
A key milestone in the development of our model involved performing a systematic literature review (SLR) of published budget impact models of lung cancer. The review identified 21 models. Findings from the SLR were presented at the Swedish Health Economics Association (SHEA) annual meeting held at the Karolinska Institute in Stockholm, Sweden on April 25, 2023. The number of identified models exceeded our initial expectations, and the time trend is positive, indicating a growing interest in this area of research. Of particular relevance for the assessment of the I3Lung AI tool, a large number of models evaluated IO therapies. While none of the identified models met our specific needs, some of them offered valuable features that have greatly contributed to the conceptualization and development of our budget impact model. Currently, a manuscript for peer-reviewed publication is under preparation.
In addition to the budget impact model, the Swedish Institute for Health Economics is also developing a cost-effectiveness model, which will be capable both of assessing the “value for money” associated with adopting the I3LUNG AI platform across participating countries and of optimizing the economic efficiency of the tool. We are in the final stages of conducting an SLR of published cost-effectiveness models of lung cancer and have identified nearly 200 relevant models. The findings from this review, which will be reported in the fall, will provide valuable insights into conceptualization of the cost-effectiveness model.
#I3LUNG #IHESweden #BIM #HorizonEurope
Article written by IHE – The Swedish Institute for Health Economics. For more information, please visit the Project section in our website or contact us.
On 29 of March -1 of April took place the ESMO – European Lung Cancer Congress in Copenhagen, Denmark, that explored the new technologies and innovations for the management of lung tumors. There were promising clinical trials showing compelling data that may pave the way for changing the way in which patients are treated today.
A recurrent concept has been the one of magic bullets to kill cancer cells. In early 1900s the physician-scientist Paul Ehrlich envisioned the concept of ‘magic bullet’, drugs that go straight to their intended cell-structural targets and could deliver a toxic drug to certain cells while sparing others, ideally the healthy one that are not affected by tumoral alterations.
More than 100 later, this concept is becoming a reality in many solid tumors with a key role in lung cancer. TROP-2, C-met, HER2, HER3 are important molecules for our cells, which are expressed at higher level than normal tissue in lung cancer tumors. A new class of drugs, called antibody-drug conjugated, has been developed in the last decades to specifically recognize these molecules on cancer cells and kill them by releasing the chemotherapy cargo they have associated. Those are Datopotamab Deruxtecan (recognizing TROP-2), Telisotuzumab Vedotin (recognizing c-MET), Sacituzumab Govitecan (recognizing TROP-2), Patritumab Deruxtecan (recognizing HER-3), Trastuzumab Deruxtecan (recognizing HER2), among others, and are revolutionizing the area of Lung Cancer.
All the above have shown interesting efficacies in phase II clinical trials in advanced non-small cell lung cancer (NSCLC) tumors, which were already pretreated with other therapies. Similarly, Patritumab Deruxtecan has shown excellent efficacies in EGFR inhibitor-resistant EGFR mutated NSCLC tumors and Trastuzumab Deruxtecan in HER2 mutant or overexpressing NSCLC tumors. Phase III clinical trials as AVANZAR, TROPION LUNG07, CARMEN LC03, Teli MET NSCLC 01, Destiny-Lung 04, MK3475-D46 among others will confirm the efficacy results previously obtained. Also, some of them will elucidate if these drugs can be positioned as preferred treatment to be administered to patients who have not received any lines of therapy before, or after the standard of chemo/immunotherapy.
Besides the ongoing trials it would be exciting to see the further development of these molecules as they may be combined with multiple treatments, with the final goal of increasing efficacy of treatment and decrease their toxicity, for example optimizing chemotherapy doses.
Following personalized medicine, KRAS G12C inhibitors continuously show efficacy results in patients with KRASG12C mutations as in the EAP -436 study. The efficacy of Amivantamab, a bispecific monoclonal EGFR and C-met antibody (meaning that is able to recognize and block molecules at the same time), was also highlighted in Osimertinib resistant tumors in the CHRISALYS-1 study. This drug was granted with accelerated approval by the FDA in 2021 for a specific population of NSCLC which are characterized by a mutation in EGFR called exon 20 insertion. Those patients are resistant to standard drug used for treating patients with EGFR-mutation, but amivantamab seems to have solved this previous unmet need and it is now standard of care for them.
Of note, approximately 2% to 3% of patients with NSCLC have EGFR exon 20 insertion mutation. It would be interesting to follow the phase III trials in naïve patients harboring EGFR mutated tumors as in the MARIPOSA trial, or for patients with EGFR Exon 20 insertions as in the PAPILLON study. It would be also interesting to see the efficacy of the drug with novel combinations and in tumors harboring C-met mutations.
A strong debate is still ongoing regarding the use of neoadjuvant immunochemotherapy (meaning before the surgery, to reduce tumor size) or adjuvant immunochemotherapy (after the surgery). The checkmate 816 phase III trial showed a 3-year EFS rate of 57% for neoadjuvant nivolumab plus chemotherapy and 43% for the chemotherapy treatment arm and an Overall survival rate of 78% versus 64% respectively. We still need to see if checkmate 816 study will change the future clinical management of resectable NSCLC. Also, it would be compelling to see the results of the coming studies evaluating neoadjuvant immunochemotherapy followed by adjuvant immunotherapy or immunochemotherapy as KEYNOTE 671, IMPOWER 030, NCT05457776, CheckMate77T among others. Last and not least, it is also interesting to evaluate if immunotherapy in the (neo)adjuvant context may yield better results compared with adjuvant immunotherapy as it was shown in advance melanoma (NCT03698019).
Cancer is a complex disease caused by mutations in DNA. Over the years, significant progress has been made in understanding its molecular basis, which has led to the development of targeted therapies. Immunotherapy is one such approach that uses the power of the patient’s immune system to fight cancer cells. Molecular biomarkers can help identify specific tumor characteristics and predict the likelihood of response to immunotherapy. However, they are not without their limitations, highlighting the need for personalized medicine.
What are Molecular Biomarkers?
Molecular biomarkers are biological molecules found in the blood, other body fluids or samples, or tumor tissues which can be used to predict the risk, progression, or response to therapy of a disease. In cancer, biomarkers are used to identify the molecular subtypes of cancer, predict prognosis, and guide treatment decisions.
Molecular Biomarkers and Immunotherapy
Immunotherapy is a type of cancer treatment that works by stimulating the patient’s immune system to recognize and attack cancer cells. While it has been a game-changer in the fight against cancer, it is not always effective for all patients, and some may experience severe side effects. In this scenario, molecular biomarkers help identify patients who are most likely to respond to immunotherapy and predict the risk of side effects.
One example of a biomarker is the programmed Death-Ligand 1 (PD-L1), a protein present on the surface of certain normal cells and at higher levels in cancer cells. PD-L1 engages with PD-1, a receptor located on T cells, a critical component of the immune system. This interaction suppresses the immune response and allows cancer cells to evade destruction by the immune system.
Limitations of Biomarkers
While molecular biomarkers can be helpful, they have certain limitations. One of the challenges with biomarkers is that they can be specific to certain types of cancer and, even within the same type of cancer, can vary between patients. For example, the PD-L1 biomarker is used to predict response to immunotherapy in lung cancer, but not all patients who express PD-L1 respond to immunotherapy.
Another limitation is that they may not provide a complete picture of the patient’s cancer, not capturing the full complexity of the cancer. This is particularly true in cases where the tumor has multiple mutations or alterations.
Lung radiography analyzed by a doctor.
The Promise of Personalized Medicine and the I3LUNG project
Personalized medicine offers a more comprehensive approach to cancer treatment by considering the unique genetic makeup of a patient’s tumor.
The I3LUNG project serves serves as an example of a personalized medicine initiative. By gathering data from over 2000 patients with metastatic non-small cell lung cancer (mNSCLC) and integrating this information with advanced technologies such as Artificial Intelligence (AI) and Machine Learning (ML), the project aims to forecast each patient’s unique response to immunotherapy. With this tool, it will be possible to design an individualized treatment plan for each patient, thereby shifting the emphasis of lung cancer care from a one-size-fits-all approach to a tailored and personalized treatment strategy, ultimately enhancing the quality of life of patients.
For more information, please visit the Project section in our website.
Artificial Intelligence in our daily lives
Artificial Intelligence (AI), despite being perceived as something straight out of science fiction, is present in our daily lives more than we think. From unlocking our phones with just a glance, to when we ask our virtual assistant for tomorrow’s weather, to using our favorite navigation app to find the less congested route… AI is there to make our lives easier and more efficient.
AI refers to the simulation of human intelligence in machines that are designed to perform tasks that would typically require human intelligence. AI systems are trained and improved over time to perform these tasks with a vast amount of data. The ultimate goal is to be able to get machines to perform certain tasks at a higher level, more accurately and efficiently.

Online Chat Bot assistant answering questions
Artificial Intelligence in the healthcare system
AI, thanks to the evolution of computer science, has made its way into the healthcare field, changing the way clinics and hospitals work.
One of the primary ways AI is supporting medical professionals is in diagnosis. With the help of AI algorithms, healthcare providers can quickly and accurately diagnose diseases, helping them to make more informed treatment decisions. AI can also assist in identifying patterns and correlations in medical data, identifying the symptoms of some diseases and matching them with specific conditions.
Another way AI is being used in clinics is for treatment planning. With AI algorithms, patient data, such as medical records, lab results and imaging studies can quickly be analyzed to determine the best treatment possibilities for a specific patient. This can help healthcare providers to personalize patient care and optimize the use of resources, improving patient experiences.
AI can also be used for predictive analytics, which involves using AI to analyze patient data and make predictions about future health outcomes. This can be particularly useful in identifying patients who are more prone to developing a particular condition, allowing intervention before the condition becomes serious.
Other ways in which AI has revolutionized the healthcare industry include:
- Increased efficiency: AI technology is able to perform repetitive and time-consuming tasks, freeing up healthcare professionals to focus on other and more critical aspects of patient care.
- Cost saving: as AI improves accuracy, it also reduces the time needed for manual analysis, optimizing resource utilization, and helping reduce the overall cost of care.
- Improved outcomes: because AI assists healthcare providers in making better treatment decisions, patients can have an improvement in their outcomes and their quality of life.
In conclusion, the integration of AI technology into the healthcare system has allowed the field of medicine to receive new and better tools to improve accuracy, efficiency and patient outcomes.

Medical technology presented on a virtual screen
I3LUNG and how AI can be used in the treatment of lung cancer
The I3LUNG project aims to provide reliable tools to patients suffering from metastatic non-small cell lung cancer (mNSCLC) through the integration of vast amounts of data from 2000 patients. This integration will be achieved by the usage of AI and Machine Learning (ML), a subfield of AI.
The project involves close collaboration with physicians and AI/ML experts with the aim of changing clinical practice in the NSCLC setting and beyond.
The ultimate goal of the I3LUNG project is to study the individual response of patients to immunotherapy, a cancer treatment that utilizes the body’s own immune system to fight and eliminate the tumor.
Not all patients respond favorably to immunotherapy, and in some cases, it can lead to toxic side effects while representing a substantial cost to the health system.
In this context, I3LUNG arises, together with AI and ML to help predict patients’ individual responses to immunotherapy, considering their specific needs, and providing them with tailored therapies that represent the best possible option for their specific situation. This will not only improve the patient’s well-being and quality of life but also help reduce the economic impact of the treatment.
Patients are the focus of I3LUNG, and as such, they will take part in the decision-making of their treatment and will be followed by psychologists during the study to collect their feedback. Understanding their thoughts on the use of AI for their cases will allow to steer the project in the best possible direction.
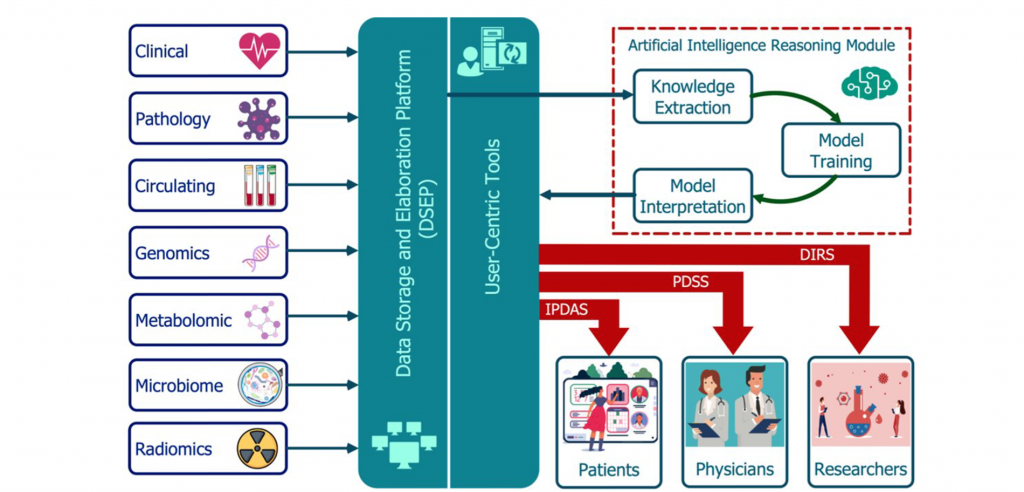
I3LUNG platform, tools, and overall structure. The AVATAR based platform and the AI/ML based tools
For more information, please visit the Project section on our website and for any doubts, contact us!
Lung cancer
Cancer is an omnipresent reality in our lives. It affects countless individuals and their families, leaving a devastating impact on all those involved. Despite its prevalence, there is still much to be learned about this disease and how to find better treatments.
The lungs are essential organs of the human body, playing a critical role in keeping us alive by controlling respiration, providing oxygen to the bloodstream, and removing carbon dioxide from the body. Unfortunately, these vital organs are also susceptible to the development of lung cancer.
Lung cancer, a malignant disease of the lung tissue, is a major global health concern. According to the World Health Organization, lung cancer has been the second most common type of cancer in 2022 and the leading cause of cancer-related deaths worldwide.
Types of lung cancer
There are two primary types of lung cancer: non-small cell lung cancer (NSCLC) and small cell lung cancer (SCLC).
NSCLC, the type of lung cancer we focus on in I3LUNG, is the most common type and is less aggressive than SCLC. It includes several subtypes, such as adenocarcinoma, squamous cell carcinoma, and large cell carcinoma.
SCLC is a less common type of lung cancer and unfortunately, it’s not detected until it has spread to other parts of the body through a process known as metastasis.
NSCLC can also metastasize, which is why early detection is critical to prevent its spread.
Demography and statistics of lung cancer
Cancer is a disease that affects people of all ages, genders, ethnicities, and races. It does not discriminate. Genetic and hormonal differences, along with various environmental exposures and other factors, can lead to disparities in cancer risk among different populations. Fortunately, the number of cases is decreasing due to people quitting smoking and advances in early detection. The majority of lung cancer cases are located in Europe, particularly in Eastern Europe (according to the World Cancer Research Fund International).
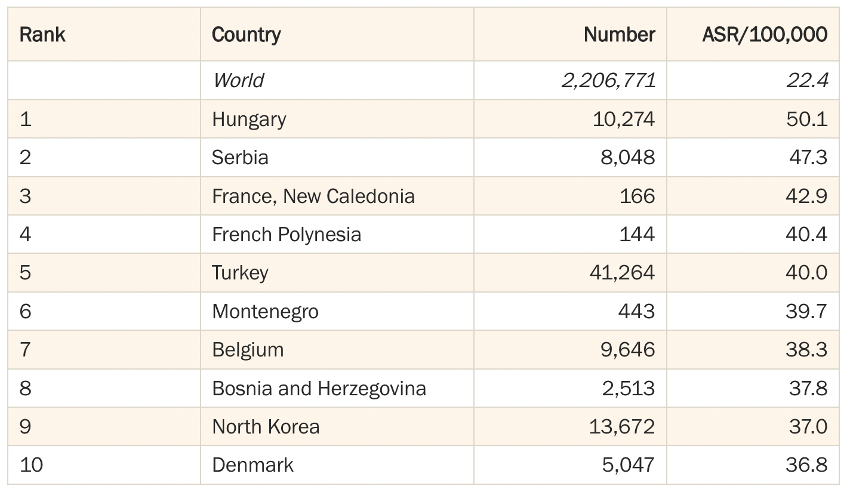
Lung cancer rates by World Cancer Research Fund International
The incidence of lung cancer increases with age, with most cases occurring in individuals around 65 years old. However, a smaller number of patients may be diagnosed at the age of 40 to 45 years old.
In contrast to decades past, when lung cancer was predominantly seen as a men’s disease, the incidence of lung cancer has risen among women. The difference in their incidence can be attributed to a variety of factors, including different smoking habits, occupational exposure to carcinogens, and genetic differences. Despite this, according to the American Cancer Society, the mortality rate for women is still lower than that of men.
The American Cancer Society reports that black men are 12% more likely to develop lung cancer than white men, while black women are 16% less likely to develop it than white women. An article by Matthew Schabath on “Racial and Ethnic Differences in the Epidemiology and Genomics of Lung Cancer” further explores the matter, mentioning that the survival rate of lung cancer patients differs according to their race and ethnicity, with white individuals having a higher survival rate than the black population. However, for small cell lung cancer (SCLC), black men are less likely to develop it than white men.
Survival rates for lung cancer vary depending on the type (SCLC being more severe than NSCLC) and the stage at which it is detected, as well as other factors. Therefore, early detection of the disease is essential to provide the patient with an effective treatment from the start.
Lung cancer prevention
The best way to prevent lung cancer is to never start smoking, or to quit it as soon as possible if you already do. Quitting smoking can drastically reduce your chances of developing the disease and improve your overall health.
Other ways to reduce the risk of having lung cancer include:
- Avoiding exposure to second-hand smoke, as it can also increase the risk of lung cancer.
- Avoiding exposure to other carcinogens: Certain occupations and industries expose workers to carcinogens such as asbestos, diesel exhaust, and certain chemicals, which can increase the risk of lung cancer. It’s important to be aware of these risks and take steps to reduce exposure.
- Eating a healthy diet and maintaining a healthy weight: Some research suggests that a diet high in fruits and vegetables may help reduce the risk of lung cancer. Being overweight or obese may also increase the risk of lung cancer.
- Exercise regularly: Regular physical activity is associated with a reduced risk of lung cancer.
It’s also worth noting that early detection and treatment of lung cancer can improve outcomes, so it’s important to be aware of the symptoms of lung cancer and to see a doctor if you have any concerns.
I3LUNG and lung cancer
At I3LUNG, we are dedicated to providing personalized treatment for patients with metastatic non-small cell lung cancer (NSCLC) by using artificial intelligence. We are committed to ensuring that patients receive the care and treatment tailored to their individual needs, to improve their quality of life during this difficult time.
For more information, please visit the Project section in our website.
After the summary done for the first day of the kick-off, let’s dive into the second and last day of the event!
Saturday, July 2nd
On the second day of the kick-off meeting the rest of the WPs were presented. The morning started with the description about the creation of the AI integrative I3LUNG platform (WP8, Integrative Platform Construction). This interface will be designed and developed to make the data in the platform available to physicians, patients and researchers. James Dolezal (University of Chicago) and Alessandro Nuara (ML Cube) have been exposing their expertise in this first morning session.
The day continued with the Economic Impact Assessment and the Ethical, Privacy and Data Protection of WP9 (Economic Impact Assessment). The objective is to analyze the socio-economic impact of I3LUNG technology and construct a user-friendly model that captures the short-term affordability and long-run cost-effectiveness. Also, the legal and ethical implication of the I³LUNG platform will be evaluated, with a particular focus on EU data protection and privacy legislation, ethics and regulations applied to AI technologies, clinical trials legislation and the medical devices legislation. Michael Willis from Swedish Institute for Health Economics and Ana Maria Correa from KU Leuven Centre for IT & IP Law (CiTiP) were the leading speaker of this session. KUL is also leading partners of WP2 (Ethical, Privacy, and Data Protection).
The final session focused on the last WP10 (Dissemination, Communication and Exploitation). Natacha Bonnet explained how MEDSIR will promote, circulate, and integrate all relevant aspects and findings from the project to the scientific and non-scientific communities. This will include participation in scientific meetings, publications on specialized scientific and technical peer-review journals, attendance to trade fairs/exhibitions, direct engagement with I³LUNG platform users, synergies with other organizations for collaborations and knowledge transfer. Another team from Italy will be in charge of the marketing and business, covering IP agreement, a market study analysis and the development of a business plan.
Finally, the I³LUNG kick-off meeting was wrapped up with a description regarding the Project Management Guidelines, a talk explaining the Next Steps by Arsela Prelaj and a final presentation about the Executive Board members.
From now on, a yearly event will be held to analyze the progression made. Feel free to reach out to the people involved to know better about the I3LUNG!

As mentioned in our previous article, the kick-off meeting on July 1-2 officially started the EU I3LUNG Project. Let’s dive deep into the first day!
Friday, July 1st
The meeting started with the introduction of the project background and an update by the principal investigator (PI) of the I3LUNG, Arsela Prelaj (Istituto Nazionale Tumori of Milan – INT). Then Melissa Fernandez from MEDSIR, the company leading WP1 (Project Management and Coordination) and managing the trial coordination, explained MEDSIR’s initial support to the I3LUNG project, drafting the proposal and its submission to the EU portal.

(From left to right: Arsela Prelaj and Melissa Fernández)
For the development of this patient decision-making treatment AI-technology, a wide range of information from around 2,200 patients, retrospective and prospective, will be captured and integrated in the AI-system (WP3, Data Capturing and Integration). Different partners from Greece (Nikolas Spathas, Metropolitan Hospital), Germany (Lung Clinic Grosshansdorf; Guido Sauter from University Medical Center Hamburg Eppendorf), Spain (Ramon Amat, Vall d’Hebron Institute of Oncology), Italy (Monica Ganzinelli and Elsa Tagliabue from Istituto Nazionale dei Tumori; Laura Brunelli from Istituto di Ricerche Farmacologiche Mario Negri) and Israel (Laila Riosman from Shaare Zedek Medical Center) explained how molecular data will be collected and analyzed in this work package. This includes collection of clinical data, pathological analysis and molecular profiling of the cancer immune environment, characterization of genomics and immune related transcriptomics, metabolomics, circulating biomarkers of response or progression, microbiome profiling and radiomics.
Patient centricity is one of the key novelties in the I3LUNG project, and for the creation of the IPDAS, different partners involved in WP4 (Patient’s quality of life – QoL – and Psychological Impact of AI on Medical Decision-Making) discussed about their specific implication in this part of the project:
- Emilia Ambrosini, from Politecnico di Milano, introduced the Sensor-based monitoring system of QoL
- Roberta Grasso from European Institute of Oncology, leading WP4, provided a general overview of how the introduction of specific psychological assessments during treatment administration can impact quality of life and decision making of patients.
- Alfonso Aguaron, representing the patients association Lung Cancer Europe, introduced specific topics related to patients recruitment.
The first day continued with more technical sessions. All the different patient-level data generated and gathered during the project have to be processed (WP5, Knowledge Extraction). The evaluable features will be extracted and delivered to the next collaborative groups for the AI algorithm design describing the real-world landscape and final model building.
- Sokol Kosta, from Aalborg University, explained how the different data gathered in the study will be integrated to generate the DIRS.
- Alexander Pearson, from University of Chicago (leading WP5) explained how to extract evaluable features on patient-level data generated from WP4 to pass forward for integrative modeling under WP6.
Francesco Trovó and Laura Pedrocchi from Politecnico di Milano, leading WP6 (Learning and Reasoning) and WP7 (Explainable AI), finally introduced the concept of AI/ML modeling and model explainability, respectively, highlighting the methods that enables humans to understand, appropriately trust, and effectively manage machine learning models while maintaining a high level of learning performance.
This intense first day ended with a pleasant dinner at a beautiful restaurant in Milan next to Duomo, where the attending representatives from the 10 WP had the chance to chat and get to know each other.



Let’s see what happened in Day 2 of the kick-off meeting!
I3LUNG is a collaborative, 5-year, 10M€ EU-funded project aiming to create an artificial intelligence (AI)-based personalized decision-making tool to aid both clinicians and patients in selecting the best lung cancer treatment plan.
The consortium behind this project comprises 16 international partners, recognized companies and institutions located worldwide, whose miscellaneous expertise in clinical trial, molecular analysis, biology, AI, psychology, ethical and business will permit the realization of this ambitious project.
The partners will collaborate on different steps for the generation of a patient avatar, able to predict progression and outcome of each individual patients, accomplished through the following:
- By supporting the physicians’ decision-making process by creating an AI-based Physician Decision Support System (PDSS) to provide an easy and ready-to-use access to predictive models, increasing care appropriateness, reducing the negative impacts of prolonged and toxic treatments.
- Facilitating important healthcare costs offsets by selecting the best personalized treatment.
- Empowering patients with an Individualized Patient Decision Aid System (IPDAS) for co-decision and reinforces research with a Data Imputation and Retrieval System (DIRS) to make securely available the data gathered during the project.

Following contract signature, the 16 partners met for a kick-off meeting on July 1 and 2 in Milan, in order to know each other face-to-face and organize the next steps and upcoming milestones of the project. This two-days intensive session was part of the project initiation phase, that brought together all the interdisciplinary teams involved to share information, align objectives and get to know each other.
Leading persons of each work package (WP) introduced their research centres, project teams, role, and responsibilities, as well as the individual and common project goals and objectives.
Let’s read in the next article how July 1st and 2nd went in details!




















